Friday, February 28, 2020
Lupine Publishers: Lupine Publishers | The Influence of Yoga on Traum...
Lupine Publishers: Lupine Publishers | The Influence of Yoga on Traum...: Lupine Publishers | Open access Journal of Complimentary & Alternative Medicine Abstract Sustaining a Traumatic Br...
Thursday, February 27, 2020
Lupine Publishers: Lupine Publishers | Subjection between Breast Canc...
Lupine Publishers: Lupine Publishers | Subjection between Breast Canc...: Lupine Publishers | Open Access Journal of Oncology and Medicine Abstract Increasing the effectiveness of antitumor the...
Groups 4 and 15 and Organotin Condensation Polymers for The Treatment of Cancers and Viruses
Groups 4 and 15 and Organotin Condensation Polymers for The Treatment of Cancers and Viruses| Lupine Publishers
Lupine Publishers| Material Science Journal
Abstract
Keywords: Cancer; Viruses; Interfacial polymerization; Brain cancer; Pancreatic cancer; Zika virus; Vaccinia virus; Breast cancer; Herpes virus
Introduction
Synthesis
R2SnX2+X-R-Y-> -(-SnR2-R-)-
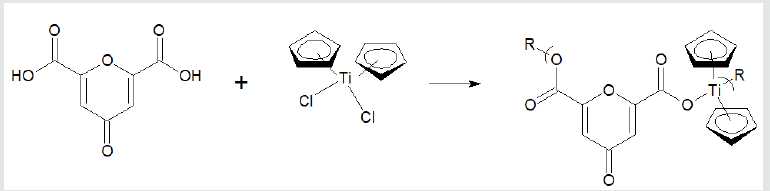
where X and Y are normally Lewis bases such as alcohols, amines, acid salts, thiols, etc. These reaction sites are often varied for a single Lewis base such as an amino acid, shown below, that has both acid and amine reactant sites. Examples of overall reaction products for each of the three condensation polymer groups are given following. Reaction between the amino acid diglycine and dimethyltin dichloride is described (Figure 1). The polymer is described as a poly (amine ester) with the organotin unit considered an organic moiety such as a methylene unit in such naming. For the Group 4 metallocenes, the reaction employing titanocene dichloride as the Lewis acid, the repeat unit for a product formed from titanocene dichloride and chelidonic acid is given (Figure 2). Finally, for reactions involving group 15 metals, the repeat unit formed from reaction between triphenylantimony dichloride and 3,5-pyridinedicarboxylic acid forming a polyester is given (Figure 3). The metal is generally located in the Lewis acid portion while the non-metal reactant is the Lewis base. In certain cases, the Lewis base portion may also contain a metal, usually iron and cobalt. The iron is present as a ferrocene while the cobalt is present as a cobaltocene [32].
Figure 1: Synthesis of organotin poly (amine esters) from
reaction of diglycine and dimethyltin dichloride where R represents
simple chain extension.
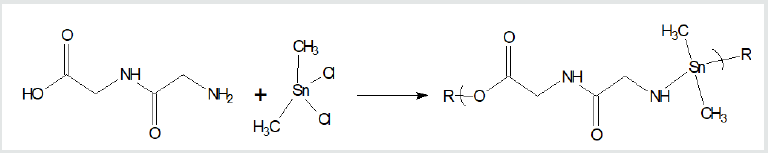

Figure 2: Synthesis of polyesters from reaction with titanocene dichloride and chelidonic acid where R represents simple chain
extension.

Cancer
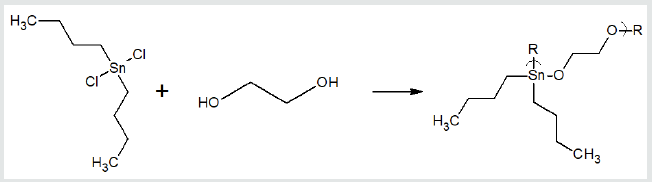
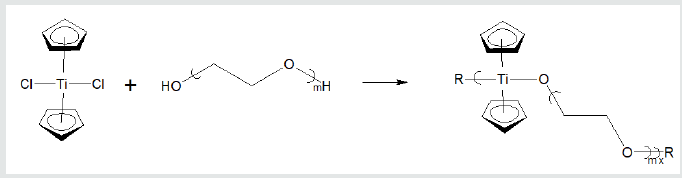
The polymers synthesized by us have shown good ability to inhibit a variety of cancer cell lines Table 1. These cell lines represent all of the major human solid tumor cell lines. These cell lines include resistant cells meaning cell lines that have shown ability to resist treatment with the traditional anticancer drugs [39] (Table 1). Inhibition depends on the metal atom present as well as the nature of the Lewis base. With respect to the metal, in general, inhibition is of the order Hf=Zr>Ti>Sn>Sb, Bi, As. Inhibition is also dependent on the specific Lewis base. A primary measure of the ability for a drug to inhibit cancer growth is the effective concentration, EC. The 50% effective concentration, EC50, is the concentration of a toxicant, drug, or antibody that induces an inhibitory response halfway between the baseline and maximum after a specified exposure time. The desired outcome is to have low EC50 values as this indicates that only a small concentration of the anti-cancer agent is needed to elicit inhibition. For the compounds described here, once inhibition begins, the slope of the dose/concentration curve is high with inhibition being total. Depending on the specific Lewis acid/base the EC50 value is typically between milligrams/mL to nanograms/mL. The metal-containing compounds are often coupled with a Lewis base that exhibits some biological activity hoping for a syngeneic effect. Drugs that have been employed as the Lewis bases include ciprofloxacin, diethylstilbestrol, cephalexin, acyclovir, thiamine, dicumarol, camphoric acid, histamine, 2-ketoglutaric acid, salicylic acid, dipicolinic acid, isomanide, glycyrrhetinic acid, phentolamine, thiodiglycolic acid. Lewis bases that themselves exhibit no ability to inhibit cancer can also exhibit good inhibition when coupled with a metal-containing moiety. These include a wide variety of diols such as ethylene glycol, Figure 4 [29,50]. Recently, water-soluble drugs possessing the metal-containing unit were synthesized [29] employing as the Lewis base poly (ethylene glycol), PEG. The resulting water-soluble polymers exhibit good inhibition of the cell lines. Figure 5 contains the reaction between titanocene dichloride and PEG forming water soluble polyethers (Figures 4 & 5).
Figure 3: Synthesis of triphenylantimony polyesters from reaction with 3,5-pyridinedicarboxylic acid where R is simple chain extension.
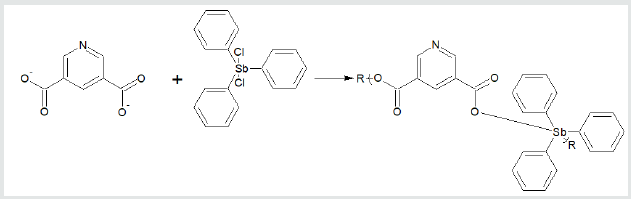

Figure 5: Formation of water-soluble polyethers from reaction of titanocene dichloride and various poly (ethylene oxides)
where R represents simple chain extension.

Viruses
From a cancer patient with ovarian cancer that had previously been treated with cytoxan, adriamycin, 5-fluorouracil, and Fur IV. From a cancer patient with ovarian cancer that had been treated with adriamycin, cyclophosphamide, and cisplatin.
Why Polymeric Drugs?
Summary
Metal-containing polymers show ability to inhibit all the major solid tumor cancers as well as important viruses. They are easily synthesized and offer physicians new drugs to attack these harmful illnesses.
Read More Lupine Publishers Material Journal Articles : https://lupine-publishers-materials.blogspot.com/
Read More Lupine Publishers blogger Articles : https://lupinepublishers.blogspot.com
Wednesday, February 26, 2020
Lupine Publishers: Lupine Publishers | Hypertrophic Cardiomiopathy in...
Lupine Publishers: Lupine Publishers | Hypertrophic Cardiomiopathy in...: Lupine Publishers | Journal of Cardiology & Clinical Research Abstract Hypertrophic cardiomyopathy (HCM) is the most co...
Lupine Publishers: Lupine Publishers | We Hear With our Brain as the ...
Lupine Publishers: Lupine Publishers | We Hear With our Brain as the ...: Lupine Publishers | Journal of Otolaryngology Research Impact Factor Abstract There are some stu...
Lupine Publishers: Lupine Publishers | Is There Any Relation Between ...
Lupine Publishers: Lupine Publishers | Is There Any Relation Between ...: Lupine Publishers | Open access journal of Complimentary & Alternative Medicine Abstract The main aim of the study ...
Thursday, February 20, 2020
Lupine Publishers: Lupine publishers | Modern Tools and Techniques fo...
Lupine Publishers: Lupine publishers | Modern Tools and Techniques fo...: Lupine Publishers | Agriculture Open Access Journal Introduction Soil salinity and brackish ground water are primary concerns for r...
Thursday, February 13, 2020
Lupine Publishers: Lupine Publishers | The Dynamics of Mounds-Cluster...
Lupine Publishers: Lupine Publishers | The Dynamics of Mounds-Cluster...: Lupine Publishers- Anthropological and Archaeological Sciences Journal Impact Factor Abstract Mounds are human made accumulation...
Wednesday, February 12, 2020
Lupine Publishers: Lupine Publishers | Non-Compacted Cardiomyopathy: ...
Lupine Publishers: Lupine Publishers | Non-Compacted Cardiomyopathy: ...: Lupine Publishers | Journal of Cardiology & Clinical Research Abstract Left ventricular non-compaction (LVNC) is a myocardial d...
Saturday, February 8, 2020
Lupine Publishers | New Materials: Current Development Under Simulation Techniques
Lupine Publishers| Modern Approaches in Material Science
Abstract
In the present short communication, a point of view on the contemporary tendencies in the development of the Science of Materials is offered. And for this, the main lines of research (personal criteria) in this area are considered, linked to problems of great importance for humanity: the care and preservation of the environment, renewable sources of energy and the health of people.
Keywords: Nanomaterials; Nanotechnology; E-skin
Introduction
In order to appreciate, in all its magnitude, the development of new materials (the totally new, the derivated, the transformed and combined ones), we need to observe through a prism of several faces, but all its converging on the plane of the climate change urgencies and the survival of the human being as a species on the planet Earth. Thus, the development of new atomic and molecular structures, the transformation of others already known, is a phenomenon closely linked to contemporary and high priority problems, such as the depletion of non-renewable sources of energy, the care and protection of the environment, and the health of people. It is possible to sustain that the emergence of modern approaches to new materials had its initial rebound in two periods of great activity: from 1821 to 1851, three decades in which it was understood at the macroscopic level and discovered the possibilities of thermoelectric; and from 1930, when it was possible to understand, from the microscopic level, thermoelectricity. This second stage led to many of the current new materials [1]. In this sense, the emergence of alternative refrigeration technologies was also decisive at the beginning of the 1990s, as a result of the combination of environmental factors and the negative evidence of global climate change. In general, the development of contemporary approaches and perspectives in the creation of new materials or the well-intentioned modification of “old” materials, is a cross-cutting phenomenon to these crucial problems of humanity, which solutions go beyond specific fields. And at the same time, in a general way, they could focus from science to suprainfim levels: nanotechnology. The manufacture of materials with great structural precision at the nanoscale has led to extremely important applications for those fields of high research demand, such as energy, environmental sciences, device technology and biomedicine. Thus, nanoarchitecture is introduced as a rising tide within the current science of nanomaterials [2]. A broad horizon, in this sense, is the discovery of graphene (“wonderful material”) and, from it, the obtaining of new two-dimensional materials such as graphyne, graphdiyne, graphone and graphane. Graphyne and graphdiyne are two-dimensional allotropes of graphene carbon with honeycomb structures. Graphone and graphane are hydrogenated derivatives of graphene. The advanced and unique properties of these new materials make them highly promising for nanoelectronics applications of next generation [3]. The already known as wonderful material has also been a bridge to reach new discoveries on principles of design and predictions of new semimetals: Dirac’s semimetals, which allow to create heterostructures from a direct layer by layer stacking, which provides an electronic coupling that facilitates a remarkable load transfer between those layers. Such structures are, apparently, very promising for the electronics of the future (Q. D. [4,5]. The material science has also managed to create crystals with optical properties that are not found in nature, whose most hopeful applications are framed in optical circuits, molecular sensors based on the resonance of surface plasmons. Comin and Manna, in their research [6] firstly explain the basic processes involved in surface plasmon resonances in nanoparticles, and later discuss the classes of nanocrystals that are particularly promising for plasmonic tunable. In the field of medicine, new materials are also playing a decisive role. For example, there are the new absorbent materials for solid phase extraction (SPE), which is the fastest growing sample preparation procedure; most usual technique in the treatment and concentration of samples before their analysis by different methods. PES are structures formed from solutions of ionic surfactants, which can be absorbed on the surfaces of active solids, resulting in sorbents capable of simultaneously extracting a wide range of analytes with an extremely varied polarity. The performance of these new SPE materials is based on molecular recognition, which mimics the selective or specific affinity of several biomolecules towards their target compounds: these absorbents include molecularly imprinted materials, immunosorbents and surfaces modified with aptamer [7]. The SPE can be considered as a giant step in the issue of obtaining samples, because the analysis of chemical compounds presents in very low concentrations in complex matrices (for example, residues and contaminants in food samples), generally requires a complex analytical approximation, involving sampling, sample preparation, isolation of analytes and qualitative and quantitative determination. In medicine, most analysts believe that the sample preparation is the Achilles heel, since it is generally time-consuming, it is prone to the introduction of contamination and it is more difficult to automate [8]. The current development of robotics is also inextricably linked to the field of medicine and novel approaches to materials. Thus, for example, the creation of an adaptable, flexible and stretchable electronic system requires the distribution of electronic products on large non-flat surfaces and mobile components. The focus of current research in this direction is marked by the use of new materials or by the intelligent engineering of traditional materials to develop new sensors, electronic components on substrates that can be wrapped around curved surfaces. Attempts are being made to achieve flexibility and elasticity in the electronic “skin”, while maintaining a reliable operation. Information about various materials that have been used in the development of flexible electronics for e-skin applications, can be found in [9]. Another current trend is the development of magnetic materials to take advantage of the magnetocaloric effect (MCE). The research focuses mainly on magnetic materials that respect the environment and their applications in heating, cooling and magnetic energy conversion technologies. However, great attention is also paid to the growing number of medical applications of the MCE, such as, for example, controllable administration and release of drugs and biomedical substances in defined places in the human body and applications of magnetic hyperthermia (cancer treatment) [10]. In the field of energy, the issue of storage is key. In studies published in 2013, electrochemical properties of materials derived from NaTi3O6 (OH) · 2H2O are revealed. The higher density and the potential for a greater speed capacity of this derivative, in comparison with the carbonaceous materials with similar voltage and reversible capacities, constitute a convincing case for its development as an anode material, both for lithium ion and sodium batteries [11]. Also, today there is a wide selection of new absorbents that can be promising for the transformation and storage of heat at low temperatures of renewable heat sources: optimization of zeolites by dealumination, further development of the aluminophosphates, the compounds “salt in the host porous matrix “, the metal-organic frames. Particular attention is focused on the chemical behavior of nano-adaptation and adjustable tuning of these materials to satisfy the demands of the appropriate cycles of heat transformation [12]. Finally, reference is made to hybrid materials, that is, materials that incorporate organic and inorganic parts. These materials have become popular in a variety of fields. The technique is not so contemporary anymore, but the incorporation of hybrid materials has given rise to a great variety of new materials and techniques to produce them. One of the most recent is the combination of the deposition of the atomic layer (ALD), which produces inorganic materials, and the deposition of the molecular layer (MLD), which produces organic materials. A variant, known as infiltration, has allowed the modification of a variety of natural and synthetic polymers with surprising results related to their general mechanical properties [13]. And what role mathematical simulation techniques has played and is playing in all the above? As has been seen, natural and artificial materials often depend on functional interfaces between organic and inorganic compounds. Examples include skeletal and biomineral tissues, drug delivery systems, catalysts, sensors, separation media, energy conversion devices and polymer nanocomposites. Current laboratory techniques are limited to monitoring and manipulating the assembly on a scale of 1 to 100nm, they are time-consuming and expensive. The confidence in the computational methods, to understand the assembly and the yield of the materials, has remarkably grown. A review of the value of the simulations compared to the experiment on the scale of 1 to 100nm, including the connections to scales of smaller length of quantum mechanics and scales of larger length of coarse-grained models, can be consulted in [14].
Conclusion
The Science of Materials, supported by its own development, and strongly “pushed upwards” by the increasing computing power, and the development of increasingly efficient and innovative simulation techniques, leads humanity towards discovery and creation of increasingly surprising materials and with a wide range of application possibilities. However, it is vitally important that such development and such possibilities do not become homicidal.
For more Lupine Publishers Open Access Journals Please visit our website: https://lupinepublishers.us/
For more Journal of Modern Approaches on Material Science articles Please Click Here: https://lupinepublishers.com/material-science-journal
To Know More About Open Access Publishers Click on Lupine Publishers: https://lupinepublishers.com/
To Know More About Open Access Publishers Click on Lupine Publishers: https://lupinepublishers.com/
Follow on Linkedin : https://www.linkedin.com/company/lupinepublishers
Follow on Twitter : https://twitter.com/lupine_online
Follow on Twitter : https://twitter.com/lupine_online
Friday, February 7, 2020
Lupine Publishers: Lupine Publishers | Somatic Mutations in Cancer-Fr...
Lupine Publishers: Lupine Publishers | Somatic Mutations in Cancer-Fr...: Lupine Publishers | Open Access Journal of Oncology and Medicine Abstract Somatic mutations have been perceived as the causal even...
Subscribe to:
Comments (Atom)
Happy Thanksgiving 2022!!!
Thanksgiving is a joyous invitation to shower the world with love and gratitude. Forever on Thanksgiving the heart wil...

-
Thanksgiving is a joyous invitation to shower the world with love and gratitude. Forever on Thanksgiving the heart wil...
-
A threat is taking over the world today. SARS-CoV-2 is a virus that spread throughout the planet, behaviorally changing world society. Human...
-
With today’s growing interest toward composite materials and their augmentation as part of integrated business from aerospace engineering, m...